Toxicology. Drug Discovery. Genomics.
Trusted by Pharma Teams. Forensically Verified.
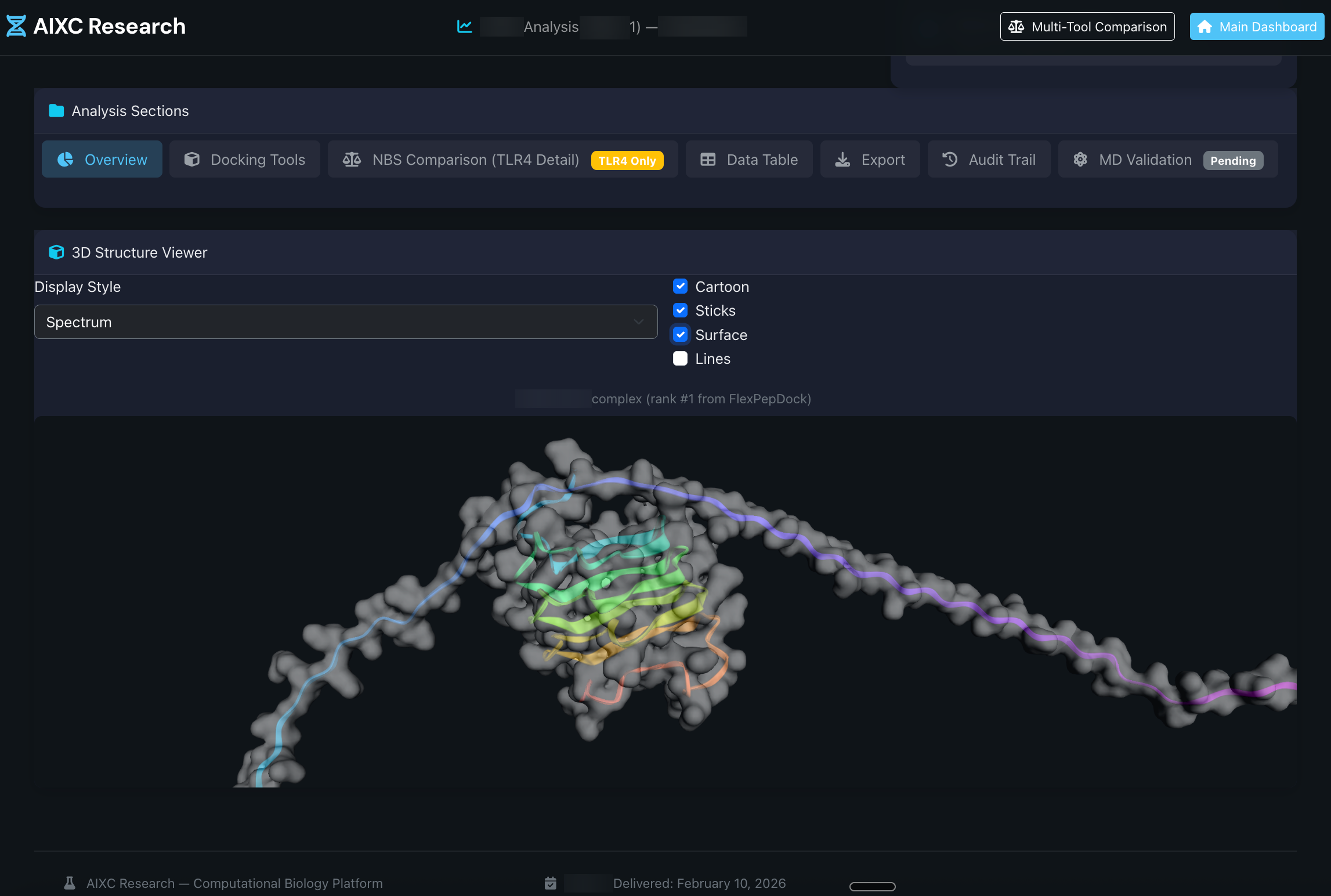
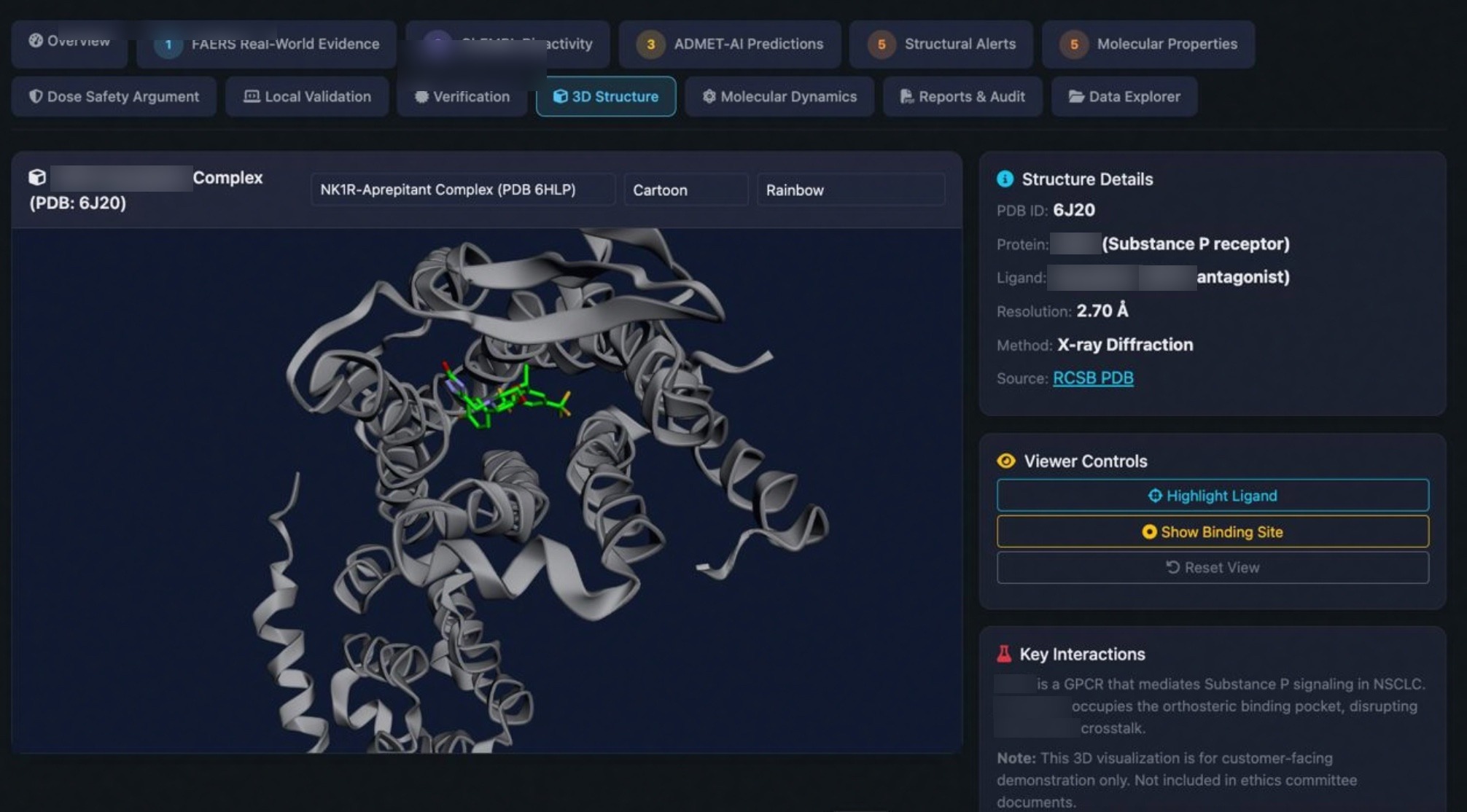
End-to-end computational drug discovery — toxicology, molecular docking, and genomics — cross-validated across 3+ independent engines at r > 0.99. Backed by 22+ peer-reviewed papers and 4 oral presentations at AAAI & NeurIPS. Per-project. 2–3 weeks.
Free · No commitment · NDA available before any data exchange